Topic: Weather And Specific Heat Of Materials
Weather And Specific Heat Of Materials
When equal masses of ice and liquid water receive the same amount of energy, without a change in state, the ice changes temperature faster than the liquid water does because the
(1) specific heat of ice is less than the specific heat of liquid water
(2) specific heat of ice is greater than the specific heat of liquid water
(3) density of ice is less than the density of liquid water
(4) density of ice is greater than the density of liquid water
What best explains why, in early spring, ice remains longer on Lake Erie than on the surrounding land areas when the air temperature is above freezing?
(1) Water has a higher specific heat than land.
(2) Energy is needed for water to evaporate.
(3) Cool winds from the surrounding land cool the ice on the lake.
(4) Air temperature does not affect water temperature.
Which group of substances is arranged in order of decreasing specific heat values?
(1) iron, granite, basalt
(2) copper, lead, iron
(3) dry air, water vapor, ice
(4) liquid water, ice, water vapor
Specific heat is used to explain why different substances
(1) sink or float in water
(2) change temperature at different rates
(3) vaporize or condense at different temperatures
(4) melt and freeze at the same temperature
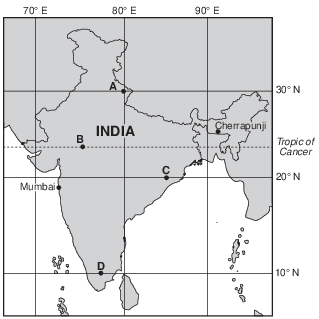
Monsoons in India
A monsoon season is caused by a seasonal shift in the wind direction, which produces excessive rainfall in many parts of the world, most notably India. Cherrapunji, in northeast India, received a record 30.5 feet of rain during July 1861. During the monsoon season from early June into September, Mumbai, India averages 6.8 feet of rain. Mumbai’s total average rainfall for the other eight months of the year is only 3.9 inches.
Monsoons are caused by unequal heating rates of land and water. As the land heats throughout the summer, a large low-pressure system forms over India. The heat from the Sun also warms the surrounding ocean waters, but the water warms much more slowly. The cooler air above the ocean is more dense, creating a higher air pressure relative to the lower air pressure over India.
The unequal heating rates of India’s land and water are caused by
(1) land having a higher density than water
(2) water having a higher density than land
(3) land having a higher specific heat than water
(4) water having a higher specific heat than land
Which factor causes the surface of Lake Ontario to cool at a slower rate than the surface of the land along the shore of the lake?
(1) Evaporating water releases more heat into the lake than into the land.
(2) Lake water has a higher specific heat than land.
(3) Water vapor cools the lake as it condenses.
(4) Sunlight passes through the top layers of the lake water.
Equal masses of granite, iron, copper, and lead are placed in sunlight. Based on specific heat, which material will warm up the fastest?
(1) granite
(2) iron
(3) copper
(4) lead
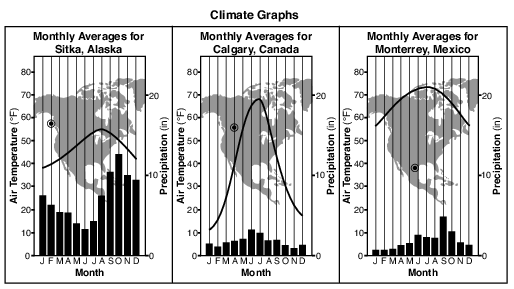
State one reason why the annual temperature range of Calgary, Canada, is greater than the annual temperature range in Sitka, Alaska. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — Calgary is surrounded by land, which has a lower specific heat than water.
• — Sitka is located near a large body of water, which has a higher specific heat than land
• materials.
• — The large body of water near Sitka moderates the temperature.
• — Calgary has a continental climate, while Sitka has a maritime climate.
• — Calgary is farther inland.

Explain why Omaha, which is farther inland, has a greater variation in temperatures throughout the year than Eureka, which is closer to the ocean. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — Omaha is surrounded by land, which has a low specific heat.
• — The Pacific Ocean moderates the temperature/climate of Eureka.
• — Large bodies of water change temperature more slowly than land does.
• — Water has a higher specific heat than land.
• — The relatively drier air around Omaha has a lower specific heat than the moist air around
• Eureka.
• Note: Do not allow credit for “Eureka is closer to water so temperatures remain constant”
• because this just restates the question without explaining the role that water plays in causing constant temperatures.
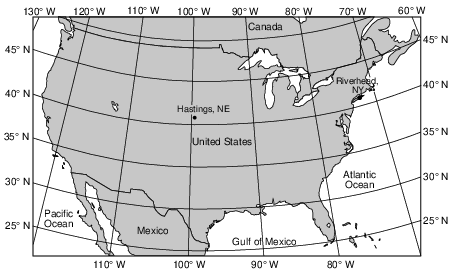
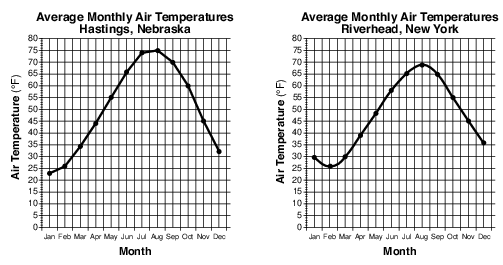
Explain why Hastings has a greater annual temperature range than Riverhead. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — Land has a lower specific heat than water and therefore causes warmer summers and
• cooler winters for Hastings.
• — Hastings is inland, where land surfaces heat up and cool down faster.
• — Riverhead is near a large body of water that moderates its temperature.
• — Land changes temperature faster than water.

Explain why the surface of Lake Erie freezes much later in the winter than the surrounding land surfaces. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — Water has a higher specific heat than land.
• — Water takes a longer time to cool than land.
• — Land surfaces cool faster.
Explain why locations near the Atlantic Ocean have air temperatures that are warmer than locations farther inland. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — The ocean changes temperature more slowly than the nearby land does.
• — Large bodies of water moderate climatic temperatures.
• — A warm ocean current is flowing nearby.
• — The water has a higher specific heat than the land does.
Great Lake Effects The Great Lakes influence the weather and climate of nearby land regions at all times of the year. Much of this lake effect is determined by the relative temperatures of surface lake water compared to the surface air temperatures over those land areas. The graph below shows the average monthly temperature of the surface water of Lake Erie and the surface air temperature at Buffalo, New York.
In an average year, four lake-effect seasons are experienced. When surface lake temperatures are colder than surface air temperatures, a stable season occurs. The cooler lake waters suppress cloud development and reduce the strength of rainstorms. As a result, late spring and early summer in the Buffalo region tends to be very sunny.
A season of lake-effect rains follows. August is usually a time of heavy nighttime rains, and much of the rainy season is marked by heavy, localized rainstorms downwind from the lake. Gradually, during late October, lake-effect rains are replaced by snows. Generally, the longer the time the wind travels over the lake, the heavier the lake effect becomes in Buffalo.
Finally, conditions stabilize again, as the relatively shallow Lake Erie freezes over, usually near the end of January. Very few lake-effect storms occur during this time period.
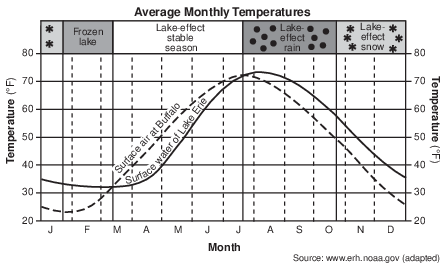
Explain why the Buffalo surface air temperatures increase faster and earlier in the year than do the surface water temperatures of Lake Erie. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — The specific heat of water is greater than the specific heat of land or dry land, so the air
• over the land heats up faster than the air over the lake.
• — More energy is required to heat up the same amount of water than to heat the same amount
• of land.
• — Air has a lower specific heat than water.
• — A lot of energy is used to melt the ice on Lake Erie.
• — Lake Erie is still covered by ice.
• — The darker land surface absorbs greater insolation.
• — Land heats up faster than water.
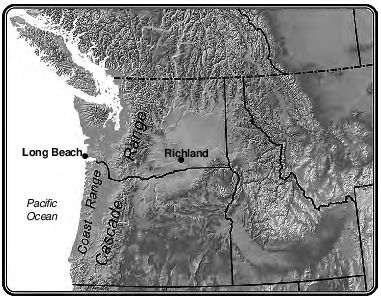
Explain why the difference between average winter and summer temperatures is smaller in Long Beach than in Richland. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — The Pacific Ocean moderates the climate of Long Beach.
• — Water has a higher specific heat and temperatures change more slowly.
• — Long Beach is near a large body of water.
• — Richland is farther inland.
• — Long Beach has a marine climate, whereas Richland’s is more continental.
• — It is located closer to a large body of water.

Explain why the winter water surface is warmer than the winter land surface. [1]
Allow 1 credit. Acceptable responses include, but are not limited to:
• — Water has a higher specific heat.
• — Water changes temperature more slowly.
• — Land has a lower specific heat.
• — It takes more energy to change the temperature of water than land.
• — Land cools off more quickly than water.
